- Home
- Details
- Registry
- RSVP
- Cleaning ears with hydrogen peroxide
- Chrome extensions font picker
- Hatoful boyfriend font
- Roller champions beta
- Ipad screen privacy protector
- Fetch a pet
- Hyperkalemia ecg findings
- Optimage vs imageoptim
- Hidden fates card list
- 3 minutes to midnight doomsday clock
- Run as admin mediakeys
- Smalland big dog
- Ocean spray
- Flow free bridges 9x9 level 25


13, 14, 15, 16 However, whether BrPh represents transient alterations in myocardial depolarization, repolarization, or any other mechanism is yet to be elucidated. The main mechanism responsible for BrS phenotype relate to abnormal patterns of repolarization and/or impulse activation at the right ventricular outflow tract (RVOT). 10, 12 Nevertheless, the clinical characteristics and prognostic impact on patients developing BrPh are poorly known. 10 Although classical hyperkalemia produces several well‐described ECG alterations, only a minority of patients develop BrPh. 3, 4, 10, 11, 12 Multiple metabolic conditions can lead to BrPh ECG the majority of cases are associated with serum K + abnormalities, hyperkalemia being the most frequent single cause.
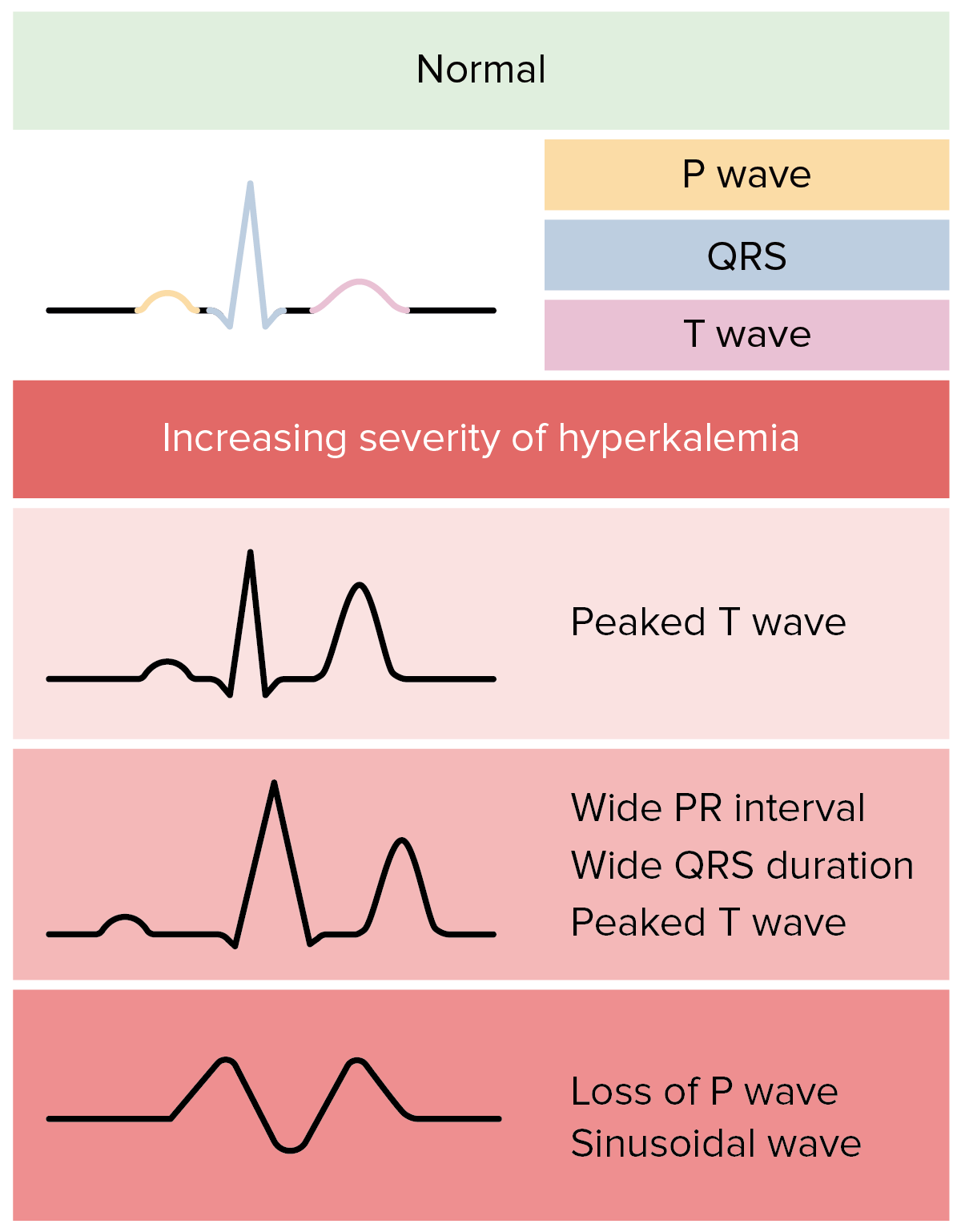
6, 9 The acquired form of Brugada‐like ECG pattern, also called Brugada phenotype (BrPh), is manifested by a Brugada ECG pattern in the presence of an identifiable underlying condition and typically disappears after its resolution. 6, 7, 8 Moreover, some environmental factors can induce ECG morphologies similar or identical to BrS in the absence of genetic mutations or ion‐channel dysfunction.
3, 4, 5 However, other studies found no clear genetic association, and minor structural abnormalities were associated with the syndrome. 3, 4 BrS has been considered as a primary electric cardiac disease caused by mutations in genes coding sodium, calcium, and potassium channels in ≈30% of patients. 1, 2 According to the 2013 consensus statement on cardiac arrhythmia syndromes and the 2016 J‐Wave syndromes expert consensus conference report, BrS is diagnosed in patients with spontaneous ST‐segment elevation (≥2 mm) with type 1 morphology in ≥1 precordial leads, particularly V1 and V2, positioned on the second and third or fourth intercostal spaces. An elevated transient outward current gradient contributed to, but was not essential for, the BrPh phenotype.īrugada syndrome (BrS) is characterized by distinctive ST‐segment elevation in the right precordial leads of the ECG, and propensity for sudden cardiac death.
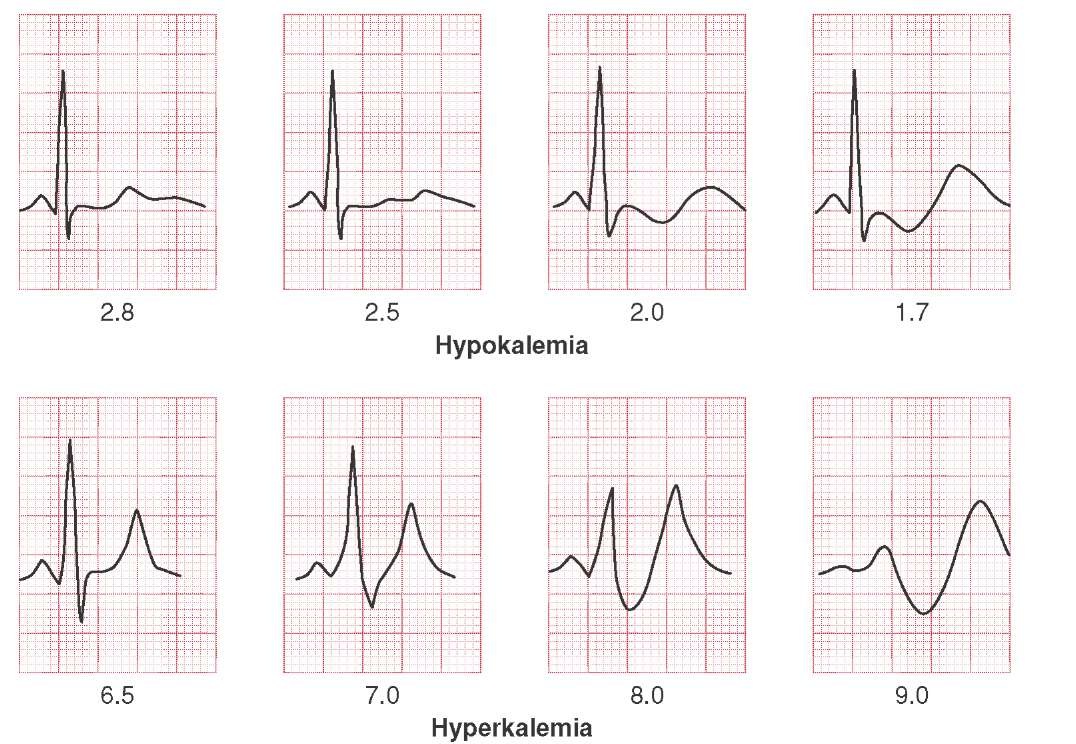
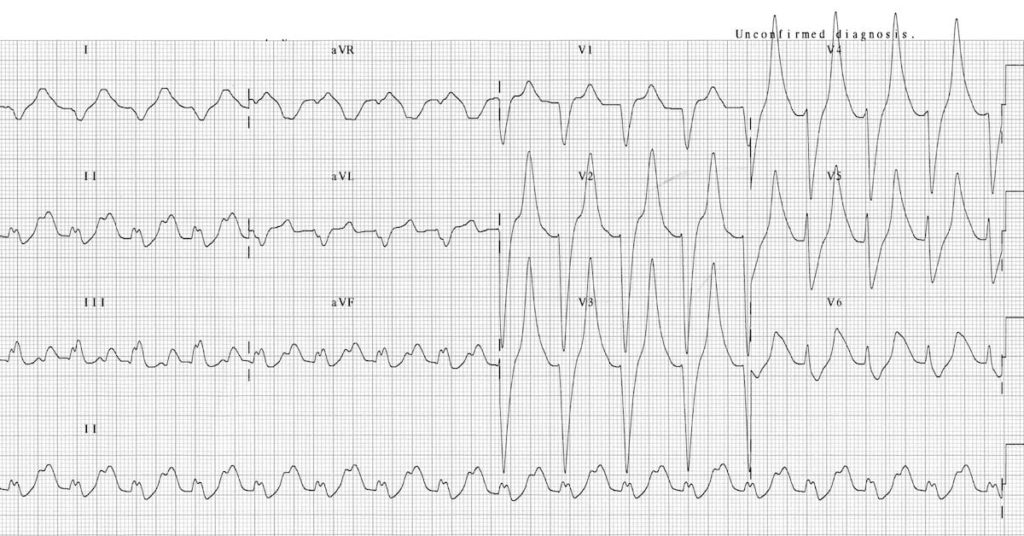
In simulations, hyperkalemia yielded BrPh by promoting delayed and heterogeneous right ventricular outflow tract activation attributed to elevation of resting potential, reduced availability of inward sodium channel conductance, and increased right ventricular outflow tract fibrosis. Multiple logistic regression analysis revealed that higher serum potassium levels (odds ratio, 15.8 95% CI, 3.1–79 P=0.001) and male sex (odds ratio, 17 95% CI, 1.05–286 P=0.045) were risk factors for developing BrPh ECG in patients with severe hyperkalemia. Six (40%) patients presented malignant arrhythmias and 6 died during admission. Most patients were admitted because of various severe medical conditions causing hyperkalemia. Over a 6‐year period, 15 patients presented severe hyperkalemia with BrPh ECG that was transient and disappeared after normalization of their serum potassium. Computer simulations investigated the roles of extracellular potassium increase, fibrosis at the right ventricular outflow tract, and epicardial/endocardial gradients in transient outward current. We prospectively identified patients hospitalized with severe hyperkalemia and ECG diagnosis of BrPh and compared their clinical characteristics and outcome with patients with hyperkalemia but no BrPh ECG. Stroke: Vascular and Interventional Neurology.Journal of the American Heart Association (JAHA).Circ: Cardiovascular Quality & Outcomes.Arteriosclerosis, Thrombosis, and Vascular Biology (ATVB).

- Home
- Details
- Registry
- RSVP
- Cleaning ears with hydrogen peroxide
- Chrome extensions font picker
- Hatoful boyfriend font
- Roller champions beta
- Ipad screen privacy protector
- Fetch a pet
- Hyperkalemia ecg findings
- Optimage vs imageoptim
- Hidden fates card list
- 3 minutes to midnight doomsday clock
- Run as admin mediakeys
- Smalland big dog
- Ocean spray
- Flow free bridges 9x9 level 25
